Division of Cancer Research
Department of Thoracic SurgeryCancer Mutomics - Exploring the Dark Matter of the Cancer Genome
In the past decade, deep sequencing approaches have revolutionized cancer genome sequencing has identified millions of somatic mutations that a tumor has acquired, but which are not present in healthy cells of same individual. However, the functional relevance of the vast majority of these mutations is unknown. We investigate the impact of mutations in cancer with an emphasis on discovering novel types of relevant mutations which we collectively refer to as "non-canonical mutations". Beyond missense and nonsense mutations altering the protein sequence, we are convinced that many other alterations can significantly effect tumorigenesis (EMBO Mol Med 2016).
Nonstop extension mutations convert a stop codon into a sense codon and thereby extending a protein at its C-terminus - and had never been studied in cancer before. We systematically compiled nonstop mutations in human cancer and provide a comprehensive database (NonStopDB). For the tumor suppressor gene SMAD4, nonstop mutations in pancreatic and colon cancer abrogate protein expression by inducing proteasomal degradation via a novel degron of ten amino acids in the added extension (Nat Cell Biol 2020). A systematic functional screen for the impact of all 2335 protein extensions emanating from nonstop / stop-loss mutations in cancer revealed that more than half of them lead to suppression of the affected protein - often by adding a detabsilizing hydrophobic C-terminus (Nat Commun 2024). Characterizing nonstop mutations in the tumor suppressor genes VHL and BAP1 in kidney cancer unraveled alternative mechanisms how nonstop mutations affect translation at different stages (Sci Adv 2025).
Synonymous mutations alter the gene and its mRNA sequence but not the protein due to the degeneracy of the genetic code. Traditionally, they have thus been viewed as "silent" mutations without a functional relavance. In a pan-cancer analysis, we have cataloged 659194 synonymous mutations in cancer at SynMICdb. Importantly, we identified relevant functions for synonymous in the strong oncogene KRAS affecting its expression and mRNA secondary structure (Nat Commun 2019).
In summary, our research thus uncovered that also so far neglected types of non-canonical mutations can have fundamental impact on cancer genes. The pioneering characterization of novel types of mutations in cancer will hence be the focus of our future research - with one main aim to dramatically increase the throughput, with which we can systematically characterize mutations in human tumor cells as endogenous model systems - to go from cancer genomics to mutomics.
Cancer Mutomics of FGFR
Fibroblast Growth Factor Receptors (FGFR1, FGFR2, FGFR3 and FGFR4) are frequently altered in human cancer - in fact, up to ten percent of all cancers across all entities harbor a genetic aberration in the FGFR family. Recently, multiple inhibitors for FGFRs (FGFRis) like Pemigatinib, Erdafitinib or Futibatinib have been approved by the FDA - and many more are currently in clinical development. FGFR translocations and high level amplifications are usually activating the pro-oncogenic FGFR signaling pathway and can often be targeted with the FGFRis. Importantly, FGFR point mutations occur frequently in cancer (e.g. in bladder cancer, lung cancer or endometrial cancer) - and at many different site with only very few hotspot regions. For the vast majority of these mutations, it is unknown whether they activate the FGFR pathway and whether they would respond to the available FGFRis.
Thus, we have established a platform for saturation mutational scanning of all four human FGFRs to test every possible point mutation for its impact on FGFR activation as well as FGFRi sensitivity or resistance. This platform allows the high throughput testing of the almost 30000 single nucleotide variants and providing a catalog of their duggability to aid in clinical decision making (Nature Genetics 2025).
For molecular tumor boards: if you have identified an FGFR point mutation in one of your patients, do not hesitate to contact us by email to obtain the results for this specific mutation from the screens which we have already performed.
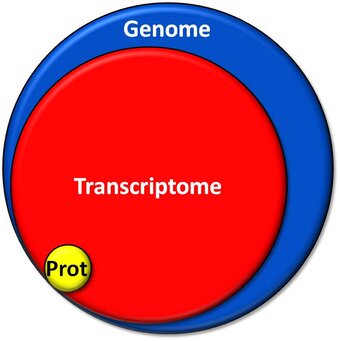
More than 70% of the human Genome (blue) is transcribed into RNA forming the Transcriptome (red), while only less than 2% of the genome are needed to produce all proteins in a human cell constituting the Proteome (yellow).
RNA Biology
A particular emphasis of our research lied on the molecular biology of RNA, its ribonucleoprotein complexes and their function in cancer.
Our research focuses on long non-coding RNAs (lncRNAs) and their role in cancer. Based on expression profiling using microarrays as well as deep RNA sequencing of the whole transcriptome, we elucidate the cellular and molecular functions of differentially regulated ncRNAs in cancer using innovative techniques like the CRISPR/Cas9 system (Nucleic Acids Res 2017), in vivo RNA Affinity Purification and our own customized siRNA and CRISPR libraries targeting specifically lung cancer-associated lncRNAs. Our RNAi & CRISPRi screens have uncovered numerous lncRNAs controlling several hallmarks of cancer including cancer cell viability, proliferation, migration and nucleotide metabolism (Nucleic Acids Res 2017, Hepatology 2018, Nat Commun 2020). We recently proposed the concept of RNA Dependence and the affiliated method as R-DeeP (Mol Cell 2019, Nat Protoc 2020).
Thesis Projects & Open Positions
We continuously welcome applications for medical doctoral theses (Dr. med.), master theses (MSc), graduate students (PhD) and postdoctoral fellows - please have a look here for open positions in Functional Cancer Genomics.
Network
We are happy to be part of the following scientific networks in Freiburg:
-
Collaborative Research Center SFB 1453
NephroGenetics
Division of Cancer Research
Department of Thoracic Surgery
Center for Translational Cell Research (ZTZ)
Breisacher Str. 115
D-79106 Freiburg

Prof. Dr. Sven Diederichs
Division Head
Phone: +49 761 270-77571
E-Mail: lung-cancer-research@uniklinik-freiburg.de



