Radiotherapy
Radiation oncology/Radiation therapy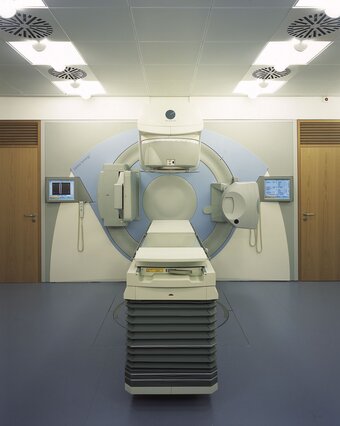
Radiotherapy (Radiation oncology/Radiation therapy) is concerned with the medical application of ionising radiation. The aim of which is to treat disease - in particular malignant but also benign - , to delay its progression or relieve discomfort. In addition to surgery and medication, radiotherapy plays an important role in the treatment of cancer. In this respect it may be employed alone, prior to, during or after surgery. Moreover it is possible to combine radiotherapy with tumouricidal pharmaceuticals (chemotherapy). It is possible to administer a chemotherapy or immunotherapy pharmaceutical that increases the tumour's sensitivity to radiation. To better protect healthy tissue radio-protective substances can also be administered. The correct treatment combination is dependent on the case and is defined in an individual treatment concept specific to the patient. Depending on the type of cancer radiotherapy is used in either a curative or palliative setting. In the case of a curative therapy the aim is to completely destroy the tumour. If this can be achieved then the patient is cured. It can also be sensible to preventatively irradiate after surgery, in order to destroy malignant cells which may remain at the surgical site. If cure of a particular cancer is no longer possible, for example if it has already metastasized, palliative radiotherapy is employed to relieve, or even eradicate, symptoms such as pain, shortness of breath, bleeding and paralysis. The well-being and quality of life of the patient would, thereby, be greatly improved.
Radiotherapy uses radiation to transmit energy that will disrupt the growth of cells in the irradiated tissue. More precisely: Radiation therapy damages the genetic material (DNA) within the cell nucleus, thereby inhibiting cell division. While healthy cells can repair the damages, tumor cells are less adaptive: They are stopped in their growth, or even destroyed.
The required dose of radiation – measured in “Gray” (Gy), an international unit named after physicist Louis Harold Gray – depends on the radiation sensitivity and the position of the tumor. The treatment plan determines for each individual patient the dosage and fractionation, i.e. the number of “single portions” for delivering the required dose. The plan must also consider the possible irradiation of healthy tissue and organs encountered by the radiation beam on its way to the tumor.
Usually the total dose is divided into several radiation sessions, or fractions. Typically fractions of 1.8 to 2.0 Gy are applied. Healthy cells need a certain amount of time to recover from radiation effects. If the next fraction is delivered after completion of this “repair work”, the patient tolerates the treatment better and has fewer side effects. Therefore radiotherapy may stretch over several weeks.
Special situations, however, in particular with high-precision radiotherapy, may provide indication of higher single doses up to 20 Gy. This is possible when such a small amount of healthy tissue is irradiated that – as in surgery – “scarring” of the affected area becomes acceptable.
A typical radiotherapy treatment, performed at a linear accelerator (linac), comprises the following steps:
- Definition of the treatment concept
An individual treatment concept is determined for every patient within the so called tumour conference, at which specialists from different medical disciplines participate: Surgeons, internists, pathologists, radiologists, psycho-oncologists, nuclear medicine physicians and palliative medicine physicians. - Patient consent
In a detailed discussion the radiation oncologist explains to the patient whether for his particular treatment whether radiotherapy will be administered as a stand alone therapy or in combination with radio-sensitising agents (radio-chemotherapy, radio-immunotherapy) and or radio-protective substances. - Preparation of the patient for radiotherapy
The position of the patient on the treatment couch will be optimally set up by means of special fixation systems. Throughout the treatment course this positioning will not be altered, in order that the same site is irradiated. In addition special positioning devices or mask systems may be constructed within our mechanical workshop. - Imaging for computer implemented planning of radiotherapy
In order to create treatment plans different imaging techniques are employed, for example computer tomography (CT) magnetic resonance imaging (MR) and positron-emission tomography (PET). This is performed in precisely the region which will be later irradiated. The radio-oncologist can distinguish between tumour and healthy tissue by means of the acquired imaging data. - Radiotherapy planning
In planning a course of radiotherapy physician and medical physicst work together closely. The radio-oncologist determines the target volume that is to be irradiated, and contours this along with organs-at-risk within the CT slices acquired in step 4. In addition he defines the therapeutic dose. A radiotherapy treatment plan and the resulting dose distribution is calculated by a medical physicist using a dedicated computer program, taking into account the relative position of the tumour, the contour of the body at the beam entrance plane and dose constraints set by the radiation-oncologist. Finally the resulting plan is evaluated by physician and physicist: only when both accept the plan and sign it off can the patient be irradiated. The treatment plan, comprising linac control parameters, is then sent to a clinical database for retrieval at the linac. After this the resulting treatment plan is checked by an independent physicist to ensure the calculated dose distribution is plausible. In addition the beam parameters for each treatment plan stored in the database are verified to ensure that data integrity. - Positioning of the patient at the therapy simulator
Before radiation therapy can begin, the patient is set up at the therapy simulator. In order to position the patient correctly for each treatment fraction small tatoos are placed on the skin which are precisely aligned with the room lasers. At this point control images are acquired, on which can be seen how and what is to be irradiated. These images compares the radiation oncologist with the treatment plan, whose accuracy is subsequently verified independently by up to two physicists. Only if everything is correct, can the actual irradiation begin. - Radiation treatment
During the course of radiation treatment patients are co-supervise by the RTTs (linac radiographers). The RTTs prepare the patients for their treatment and operate the linac. In view of this they work closely with physicians, medical-physicists and staff from the mechanical workshop. The actual irradiation does not take long: depending on plan complexity and the administered dose this ranges from a few seconds to several minutes. - Verification during treatment
During the treatment the accuracy of the radiation application is regularly verified, for example by CT or x-ray imaging directly at the linac. In this manner the position of tumours, which move due to breathing or heart beat can be precisely determined and if necessary the position of the patient on the treatment couch can be appropriately adjusted. - Doctor's discussion
During the radiation treatment regular discussions with a physician take place. Futhermore if questions or problems arise the patients can turn to the treating radio-oncologist. - End of radiotherapy
At the end of the course of radiation a detailed discussion with the treating physician takes place. During this discussion, amongst other things, it will be clarified whether the treatment is completed or whether further therapeutic procedures, such as radio-chemotherapy, are necessary. - Follow-up examination
About 4 to 6 weeks after the end of radiotherapy the patient comes to the clinic for the first follow-up examination. A physician thoroughly examines the patient to determine how well the radiation treatment worked and whether the tumour is now under control or healed. In addition the physician checks whether side-effects or late effects are present, in order to begin appropriate procedures. The legally prescribed follow-up last several years and is performed in close cooperation with the appropriate specialists and the patient's GP.
When creating radiotherapy treatment plans different imaging methods are employed, for example computer tomography (CT), magnetic resonance imaging (MR) and positron-emission tomography (PET). Like a surgeon, who must separate a tumour from the surrounding healthy tissue, the radiation-oncologist must unequivocally distinguish the tumour affected structures from the healthy tissue. The correct contouring of the target volume and organs-at-risk is decisive for the outcome of the radiotherapy treatment. Due to this several clinical studies are currently running with the Department of Radiation Oncology which have the potential to further improve the possibilities of radiotherapy.

Prof. Dr. med. Anca-L. Grosu
Chair of Department of Radiation Oncology
Registration and Information
Ambulatory healthcare center
Telefon +49 (0) 761 270-94620
Telefax +49 (0) 761 270-95820
strahlenheilkunde.anmeldung@uniklinik-freiburg.de
Private patients healthcare center
Telefon +49 (0) 761 270-94730
Telefax +49 761 270 94720
Office manager
Telefon +49 (0) 761 270-94610
Telefax +49 (0) 761 270-94720
britta.cousin@uniklinik-freiburg.de
