Selected Projects in Pillar 2: Diagnostic Innovations and Molecular Prevention
Under Diagnostic Innovations and Molecular Prevention, the selected projects focus on advanced diagnostics, early detection, and biomarker-driven clinical decision-making:
This project aims to translate immune based diagnostics into primary liver cancers, particularly hepatocellular carcinoma (HCC) and biliary tract cancer (BTC), cancers with high mortality despite recent advances in immunotherapy. Although immune checkpoint inhibitors are now standard first-line treatments, only a minority of patients benefit from these therapies, highlighting a critical need for predictive biomarkers. Current clinical practice likely results in both overuse and underuse of immunotherapy depending on disease stage. The project is based on the hypothesis that the spatial organization and composition of the tumor immune microenvironment determine responsiveness to immunotherapy and can be leveraged to guide personalized treatment decisions. We have demonstrated that high-dimensional profiling approaches—combining mass cytometry, spatial analysis, and digital pathology—can identify key immune cell populations, such as exhausted and tissue-resident T cells (Barsch et al., J Hepatol 2022), and define spatial immune patterns that correlate with treatment outcomes (Salié et al., Gut 2025). We also uncovered mechanistic insights into immunotherapy response, including the role of IL-21 signaling in enhancing CD8 T-cell activity (Zhang et al., Nat. Immunol 2025).
In the proposed work, these findings will be translated into a clinically applicable immune classification and scoring system. First, the approach will be extended to biliary tract cancer to characterize its immune landscape and identify spatial features associated with clinical outcomes. Second, the classification system will be validated in independent HCC patient cohorts undergoing different immunotherapy regimens, including both advanced and early-stage disease, to determine its predictive value across treatment settings. Finally, the project aims to simplify the complex multiomics-based framework into a practical tool for routine clinical use by developing a reduced marker panel compatible with standard pathology techniques and creating standardized reporting formats for tumor boards. Overall, the ambition of the project is to bridge the gap between experimental discovery and clinical implementation by delivering an integrated, immune-based diagnostic pipeline that supports personalized therapy decisions and can contribute to improve outcome for patients with liver cancer.
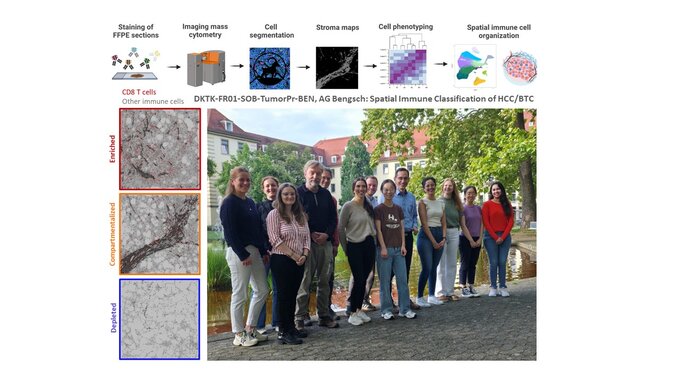
Top: Workflow illustrating Imaging Mass Cytometry data preparation and analysis steps. Bottom left: Simplified spatial immunotypes based on CD8 T cells and Collagen staining of a HCC tissue allowing the classification of the tumor microenvironment in 3 immunotypes: immune-enriched/compartmentalized/depleted. CD8 T cells are depicted in red and other cells in grey. Bottom right: Picture of AG Bengsch at the Uniklinik Freiburg
Primary central nervous system lymphoma (PCNSL) is an aggressive extranodal lymphoma characterized by marked clinical heterogeneity and poor outcomes in a substantial proportion of patients, with relapse rates of up to 50% despite intensive methotrexate-based therapies. Current clinical and radiological risk stratification tools lack sufficient sensitivity and specificity, resulting in a largely uniform treatment approach that exposes many patients to unnecessary toxicity.
This proposal aims to establish ultrasensitive circulating tumor DNA (ctDNA) profiling as a robust, noninvasive biomarker for response assessment and risk-adapted treatment stratification in PCNSL. Building on prior work demonstrating the feasibility and prognostic relevance of ctDNA, the project leverages biospecimens and clinical data from the large, international phase III trials OptiMATe and PRIMA-CNS of the German Cooperative Study Group CNS Lymphoma (GCSH-CNSL), which provide a unique and comprehensive resource of longitudinal tumor tissue, plasma, and cerebrospinal fluid (CSF) samples. The project is structured around three central aims: (i) to define the mutational landscape of PCNSL tumors, (ii) to perform ultrasensitive longitudinal ctDNA monitoring across multiple disease time points in plasma and CSF, and (iii) to determine the optimal timing and biological compartment for ctDNA-based prognostication. Methodologically, the project combines advanced next-generation sequencing technologies with a well-curated, prospectively collected biobank, enabling high-resolution longitudinal analyses. The translational design ensures a direct link between biomarker development and clinical application.
Overall, this work addresses a critical unmet clinical need by enabling precise, biomarker-guided treatment strategies in PCNSL. Successful implementation could reduce overtreatment, improve patient outcomes, and pave the way for ctDNA-guided clinical trials.
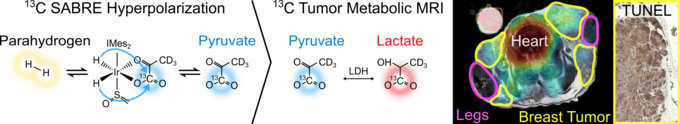
Altered metabolism is a hallmark of cancer and provides valuable information for tumor characterization, therapy selection, and early assessment of treatment response. However, the ability to non-invasively probe metabolic pathways in real time remains limited in current clinical practice.
In this project, we explore new approaches based on parahydrogen-induced signal enhancement to enable rapid and flexible metabolic magnetic resonance measurements. These quantum-enabled methods have the potential to substantially increase sensitivity and throughput compared to conventional techniques, thereby opening new opportunities for translational cancer research.
A central goal is to investigate strategies for scaling hyperpolarized contrast generation to volumes suitable for studies in large-animal models, representing an important step toward future clinical translation. In parallel, the project evaluates the feasibility to study dynamic responses to therapeutic interventions.
The project aims to generate key knowledge on how next-generation hyperpolarized magnetic resonance methods can be implemented in translational oncology. The long-term vision is to contribute to more accessible and informative metabolic imaging tools that support personalized cancer diagnostics and treatment strategies.