AG Dr. Gentle
Dr. Ian Gentle
| Telefon Büro | +49 761 270-83622 |
|---|---|
| Fax | +49 761 270-83703 |
| ian.gentle@uniklinik-freiburg.de |
CV
| 2002 – 2006 | PhD – University of Melbourne, Biochemistry and Molecular Biology Department and Bio21 Molecular Science and Biotechnology Institute, Melbourne, Australia. |
| 2006 – 2008 | Postdoc – LaTrobe University, Biochemistry Department, Melbourne, Australia. |
| 2008 – 2010 | National Health & Medical Research Council (NH&MRC) Postdoctoral Fellow – LaTrobe University, Biochemistry Department, Melbourne, Australia. |
| 2010 – 2012 | European Molecular Biology Organisation (EMBO) Long Term Fellow – Universitätsklinikum Freiburg, Department für Medizinische Mikrobiologie und Hygiene, Institut für Medizinische Mikrobiologie und Hygiene |
| 2012 – present | Postdoc – Universitätsklinikum Freiburg, Department für Medizinische Mikrobiologie und Hygiene, Institut für Medizinische Mikrobiologie und Hygiene. |
Who is who?
| Name | Postition | Telefon | |
|---|---|---|---|
| Dr. Ian Gentle | AG-Leiter | +49 761 270- 83622 | ian.gentle@uniklinik-freiburg.de |
| Sylwia Gradzka | PhD-Student | +49 761 270- 83658 | sylwia.gradzka@uniklinik-freiburg.de |
| Arlena Metz | BTA | +49 761 270- 83657 | arlena.metz@uniklinik-freiburg.de |
Publikationen
- Goodall KJ, Finch-Edmondson ML, van Vuuren J, Yeoh GC, Gentle IE, Vince JE, Ekert PG, Vaux DL, Callus BA. Cycloheximide Can Induce Bax/Bak Dependent Myeolid Cell Death Independently of Multiple BH3-Only Proteins. PLoS one. 2016 Nov 2;11(11):e0164003
- Waguia Kontchou C, Tzivelekidis T, Gentle IE, Häcker G. Infection of epithelial cells with Chlamydia trachomatis inhibits TNF induce apoptosis at the level of receptor inernalizatin while leaving non-apoptotic TNF-signalling intact. Cell Microbiol. 2016 Nov;18(11):1583-1595
- Gentle IE, Moelter I, Lechler N, Bambach S, Vucikuja S, Häcker G, Aichele P. Inhibitor of apoptosis proteins (IAPs) are required for effective T cell expansion/survival during anti-viral immunity in mice. Blood. 2013;Dec 12 [Epub ahead of print].
- Vince JE, Wong WW-L, Gentle I, Lawlor KE, Allam R, O'Reilly L, Mason K, Gross O, Ma S, Guarda G, Anderton H, Castillo R, Häcker G, Silke J, Tschopp J. Inhibitor of Apoptosis Proteins Limit RIP3 Kinase-Dependent Interleukin-1 Activation. Immunity. 2012;36:215–227.
- Gentle IE, Wong WW, Evans JM, Bankovacki A, Cook WD, Khan NR, Nachbur U, Rickard J, Anderton H, Moulin M, Lluis JM, Moujalled DM, Silke J, Vaux DL. In TNF-stimulated cells, RIPK1 promotes cell survival by stabilizing TRAF2 and cIAP1, which limits induction of non-canonical NF-k B and activation of caspase-8. J Biol Chem. 2011;286:13282-91.
- Dagley MJ, Gentle IE, Beilharz TH, Pettolino FA, Djordjevic JT, Lo TL, Uwamahoro N, Rupasinghe T, Tull DL, McConville M, Beaurepaire C, Nantel A, Lithgow T, Mitchell AP, Traven A. Cell wall integrity is linked to mitochondria and phospholipid homeostasis in Candida albicans through the activity of the post-transcriptional regulator Ccr4-Pop2. Mol Microbiol. 2011;79:968-89.
- Lluis JM, Nachbur U, Cook WD, Gentle IE, Moujalled D, Moulin M, Wong WW, Khan N, Chau D, Callus BA, Vince JE, Silke J, Vaux DL. TAK1 is required for survival of mouse fibroblasts treated with TRAIL, and does so by NF-kB dependent induction of cFLIPL. PLoS One. 2010;5:e8620. Erratum in: PLoS One. 2010;5(12) doi: 10.1371/annotation/4e35cd59-c68f-4020-addb-18eb896112c5.
- Wong WW, Gentle IE, Nachbur U, Anderton H, Vaux DL, Silke J. RIPK1 is not essential for TNFR1-induced activation of NF-kB. Cell Death Differ. 2010;17:482-7.
- Vince JE, Pantaki D, Feltham R, Mace PD, Cordier SM, Schmukle AC, Davidson AJ, Callus BA, Wong WW, Gentle IE, Carter H, Lee EF, Walczak H, Day CL, Vaux DL, Silke J. TRAF2 must bind to cellular inhibitors of apoptosis for tumor necrosis factor (tnf) to efficiently activate NF-kB and to prevent tnf-induced apoptosis. J Biol Chem. 2009;284:35906-15.
- Alcock FH, Grossmann JG, Gentle IE, Likić VA, Lithgow T, Tokatlidis K. Conserved substrate binding by chaperones in the bacterial periplasm and the mitochondrial intermembrane space. Biochem J. 2008;409:377-87.
- Gentle IE*, Perry AJ*, Alcock FH, Likić VA, Dolezal P, Ng ET, Purcell AW, McConnville M, Naderer T, Chanez AL, Charrière F, Aschinger C, Schneider A, Tokatlidis K, Lithgow T. Conserved motifs reveal details of ancestry and structure in the small TIM chaperones of the mitochondrial intermembrane space. Mol Biol Evol. 2007;24:1149-60.
- Burri L, Vascotto K, Gentle IE, Chan NC, Beilharz T, Stapleton DI, Ramage L, Lithgow T. Integral membrane proteins in the mitochondrial outer membrane of Saccharomyces cerevisiae. FEBS J. 2006;273:1507-15.
- Burri L, Strahm Y, Hawkins CJ, Gentle IE, Puryer MA, Verhagen A, Callus B, Vaux D, Lithgow T. Mature DIABLO/Smac is produced by the IMP protease complex on the mitochondrial inner membrane. Mol Biol Cell. 2005;16:2926-33.
- Gentle IE, De Souza DP, Baca M. Direct production of proteins with N-terminal cysteine for site-specific conjugation. Bioconjug Chem. 2004;15:658-63.
- Gentle I, Gabriel K, Beech P, Waller R, Lithgow T. The Omp85 family of proteins is essential for outer membrane biogenesis in mitochondria and bacteria. J Cell Biol. 2004;164:19-24.
* Indicates equal contribution
- Gentle IE, Silke J. New perspectives in TNF-R1-induced NF-kB signaling. Adv Exp Med Biol. 2011;691:79-88.
- Gentle I & Nachbur U. Scorched Earth or Viral Birth? The Open Cell Signaling Journal. 2011;3:35–41. Review.
- Gentle IE, Burri L, Lithgow T. Molecular architecture and function of the Omp85 family of proteins. Mol Microbiol. 2005;58:1216-25. Review.
Forschungsprojekt
Inhibitor of Apoptosis Proteins (IAPs) in regulation of immune signalling
Summary:
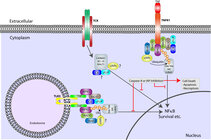
We are interested in both Toll Like Receptors (TLRs) and T Cell Receptor (TCR) complex assembly and signalling regulation. We are specifically interested in the role Inhibitor of Apoptosis Proteins (IAPs) and RIP kinases play at these receptors. Loss of IAP function in either case leads to cell death and disruption in downstream signalling pathways. RIP kinases are able to activate cell death in the absence of caspase activity in a process known as necroptosis, and can be activated by both TLR and TCR activation as well as TNF receptor activation. RIP kinases are targets of IAP mediated ubiquitylation, which regulates their activity. IAP mediated ubiquitylation of RIPK1 and other substrates recruits ubiquitin binding complexes TAB/TAK, LUBAC and IKK in order to activate downstream transcriptional responses such as NF-kB. This can be seen summarized in the simplified figure below.
Loss of IAP function can lead to problems in control of infection and potentially pathological inflammation. We use various in vitro models such as gene deficient cell lines and also mouse models of LCMV infection to study how IAPs and RIP kinases can regulate the decision of these receptors to trigger cell death or drive inflammation. We are also interested in how signals from these receptors are switched off. Once activated these receptors must be later switched off in order to avoid prolonged activation leading to inflammation. Loss of autophagy has been shown to lead to severe inflammatory responses in response to triggers such as TLR4 activation by LPS and mutations in autophagy genes have been linked to Crohns disease. We are studying how processes such as autophagy may be involved in silencing receptors by degradation. And how IAP mediated ubiquitylation may be regulating this aspect of signalling.