Cardiac Optogenetics
The Optogenetics Group develops genetically encoded, light-activated tools to study cardiac physiology, from single cell-signalling to whole-heart mechano-electric activity. At present, research activities are focussed on three main research topics:
(1) the design and implementation of novel light-gated ion channels for modulating cardiomyocyte activity,
(2) the study of heterocellular coupling between cardiomyocytes and cardiac interstitial cells, and
(3) the investigation of neuronal control of heart function.
Novel light-gated ion channels
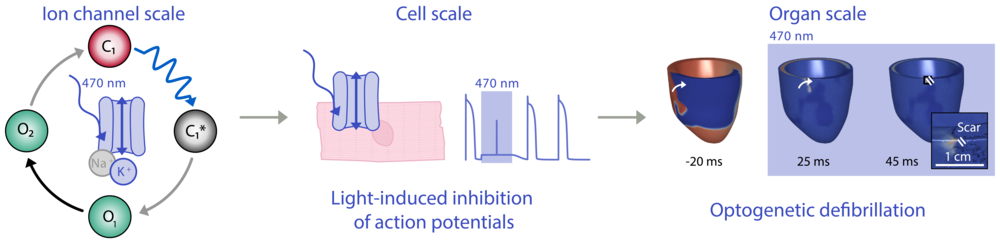
Over the last decade, we have contributed to the development and functional testing of novel light-gated ion channels, including the newly found groups of light-gated chloride and potassium channels. Using patch-clamp measurements, contraction tracking, and computational modelling, we systematically identify biophysical parameters that determine effects of channel activation on cardiomyocyte activity. At present, we study how the ion selectivity of light-gated channels determines their potential to terminate cardiac re-entry arrhythmia. This project is currently funded by the Medical Scientist Programme of the DGK.
Characterising light-gated K+ channels across scales. Left: Simplified photocycle model. Middle: Inhibition of action potentials in cardiomyocytes. Right: Simulation of K+-channel mediated defibrillation of re-entry arrhythmia in human ventricle.
Heterocellular coupling between cardiomyocytes and interstitial cells
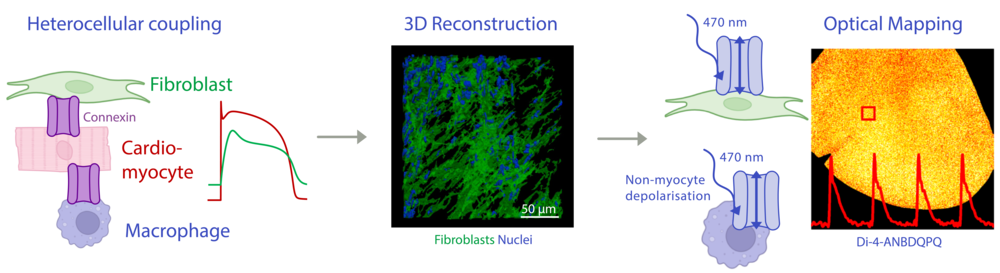
In native myocardium, cardiomyocytes are surrounded by interstitial non-myocytes that shape the electromechanical activity of the heart. We are interested in the bidirectional biophysical crosstalk between muscle and interstitium, and how these interactions affect cardiac function in physiological and pathophysiological conditions. To study this, we use optical mapping and optogenetic manipulation, tissue clearing, 3D imaging, and in silico models. These activities are embedded within the newly established research group FOR6051.
Effects of heterocellular electrotonic coupling between cardiomyocytes and interstitial non-myocytes. Left: Schematic overview. Middle: 3D reconstruction of fibroblast network in ventricular scar tissue. Right: Functional experiments combine optogenetic stimulation of non-myocytes with dye-based optical mapping of membrane voltage.
Neuronal control of heart function
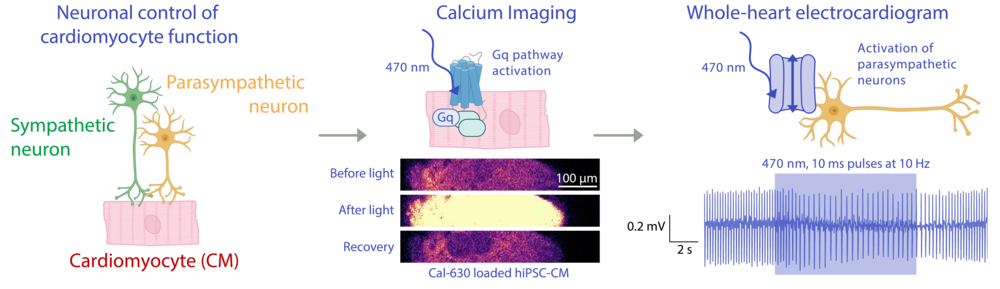
Using custom-developed optogenetic approaches, we study how autonomous activity shapes cardiac electrophysiology and calcium dynamics, and whether dysregulation of balanced neuronal control may promote cardiac arrhythmogenesis. We bridge from microscale characterisation of neuro-cardiac junctions to whole-heart electrophysiology, and also assess downstream integration of selected G-protein pathways in cardiomyocytes and non-myocytes. These projects are funded by FOR5807 and an ERC Synergy Grant.
Optogenetic interogation of neuron-cardiomyocyte crosstalk. Left: Scheme depicting the cell types involved. Middle: Optogenetic activation of the Gq-protein signalling pathway visualised by calcium imaging. Right: Optogenetic stimulation of parasympathetic neurons decreases heart rate.
- Ohnemus S, Tillert L, De Zio R […] Leemisa AN […] Kohl P, Timmermann V, Schneider-Warme F, Vierock J. Experimentally informed, quantitative photocycle model of the light-gated potassium channel WiChR. Biophys J 2026/in press
- Spreen A, Sadanandan N, Schneider MW […] Leemisa AN, De Zio R […] Heer J […] Schneider-Warme F, Bernal Sierra YA, Seifert R. Optogenetic silencing by combining a rhodopsin cyclase with an engineered cGMP-gated potassium channel. Sci Adv 2025/11:eadx1195
- Stefanovska D, Sassu E, Tekman M […] Perez-Feliz S […] Schneider-Warme F […] Preissl S, Hortells L. Fibroblast growth factor signaling induces a chondrocyte-like state of peripheral nerve fibroblast during aging. Nat commun 2025/16:10020
- Chan A, Greiner J, Marschhäuser L, Brennan TA, Perez-Feliz S […] Klesen A […] Rog-Zielinska E, Kohl P, Schneider-Warme F, Grün D. Spatio-temporal dynamics of the cardio-immune niche during lesion repair. Nat Cardiovasc Res 2025/4:1550-1572
- Simon-Chica A, Klesen A, Emig […] Greiner J […] Rog-Zielinska EA, Ravens U, Kohl P, Schneider-Warme F, Peyronnet R. Piezo1 stretch-activated channel activity differs between murine bone marrow-derived and cardiac tissue-resident macrophages. J Physiol 2024/602:4437-4456
- Sassu E, Tumlinson G, Stefanovska G, Fernández MC, Iaconianni P, Madl J, Brennan TA, Koch M, Cameron BA […] Ravens U, Schneider-Warme F, Kohl P, Zgierski-Johnston CM, Hortells L. Age-related structural and functional changes of the intracardiac nervous system. J Mol Cell Cardiol 2024/187:1-14
- Vierock J, Peter E, Grimm C […] Casini M […] Peyronnet R, Schneider-Warme F […] Hegemann P. WiChR, a highly potassium-selective channelrhodopsin for low-light one- and two-photon inhibition of excitable cells. Sci Adv 2022/8:eadd7729
- Simon-Chica A, Fernández MC, Wülfers EM, Lother A, Hilgendorf I, Seemann G, Ravens U, Kohl P, Schneider-Warme F. Novel insights into the electrophysiology of murine cardiac macrophages: relevance of voltage-gated potassium channels. Cardiovasc Res 2022/118(3):798-813
- Kopton RA [...] Moss R, Zgierski-Johnston CM [...] Kohl P, Quinn TA, Schneider-Warme F. Cardiac Electrophysiological Effects of Light-Activated Chloride Channels. Front Physiol 2018/9:1806
- Bernal Sierra YA*, Rost BR*, Pofahl M* [...] Kopton RA [...] Kohl P, Baier H, Schneider-Warme F [...] Schmitz D. Potassium channel-based optogenetic silencing. Nat Commun 2018/9:4611 (*equal contribution)
Team

Dr. Franziska Schneider-Warme
Head of section

Jan Rebers
E-Mail: jan.rebers@uniklinik-freiburg.de

Roberta De Zio, PhD
Guest Scientist

Ana Simón Chica, PhD
Guest Scientist