Translational Research
This section connects the basic research of the Institute for Experimental Cardiovascular Medicine with the clinical application at the University Heart Center.
Preventing Ischemia-Reperfusion Arrhythmias
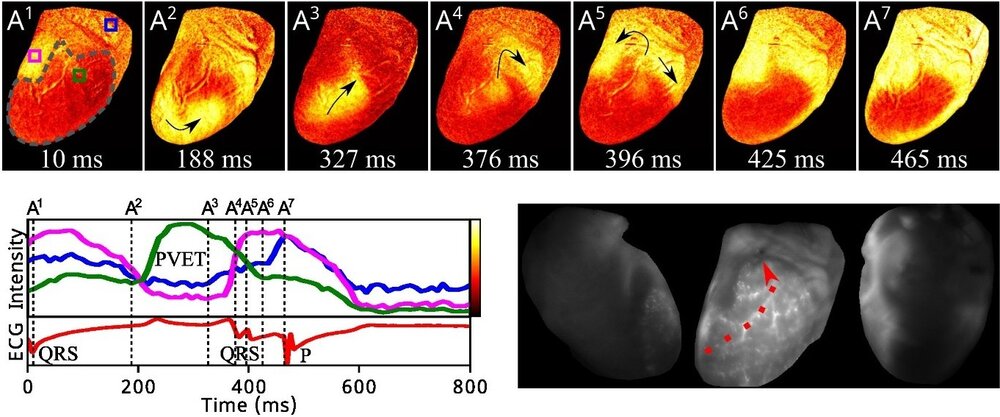
A heart attack typically occurs due to blockage of a coronary artery, leading to an ischaemic area. Removal of this block is necessary to restore tissue health, however this paradoxically can lead to additional damage including arrhythmias. We have recently identified a novel mechanism of these ischaemia-reperfusion arrhythmias, which we call perivascular excitation tunnelling (Chleilat et al.). Here, the myocardium closest to the main branch of the blocked coronary vessel returns to excitability first, leading to a conduction highway through the previously ischaemic tissue. In a BMFTR-funded project, we are developing a novel reperfusion catheter together with OSYPKA GmbH, that in pilot experiments completely prevents arrhythmia occurrence. Ongoing work in this project involves optimizing the catheter design, its use and handling, and the application of it in isolated hearts and in vivo with the long-term goal of preventing arrhythmias in patients and improving outcomes post heart attack.
This project is led by Callum Zgierski-Johnston.
Fig. 1: Arrhythmia following reperfusion. A1-A7 frames showing cardiac excitation travelling through the previously ischaemic zone (grey dashed lines) leading to re-excitation of the whole heart. Bottom left: intensities from regions of interest shown in A1 and the corresponding electrocardiogram demonstrating the inverted excitation (QRS wave followed by P). Bottom right: image following perfusion with fluorescent microspheres showing the main branch of the perfused coronary artery.
Improving Ablation
Atrial fibrillation is one of the most common cardiac arrhythmias. Among the latest strategies to treat it is pulsed-field ablation, which uses brief high-voltage electrical pulses to create irreversible pores in cardiac cells, thereby interrupting conduction and stopping the arrhythmia.
As part of a BMFTR-funded project, we collaborate with industry partners developing generators and catheters to better understand lesion formation and the parameters that determine lesion shape. Using translational models and human tissue from the CardioVascular BioBank, we assess pulsed-field ablation efficiency through a combination of approaches, including optical mapping, live/dead cell staining, and histological techniques.
By integrating experimental research with artificial intelligence, in collaboration with the Karlsruhe Institute of Technology, our long-term goal is to tailor ablation protocols to specific tissue properties and ultimately to individual patients.
The first results of this project have been recognised at the annual meeting of the German Cardiac Society (DGK).
This project is led by Rémi Peyronnet.
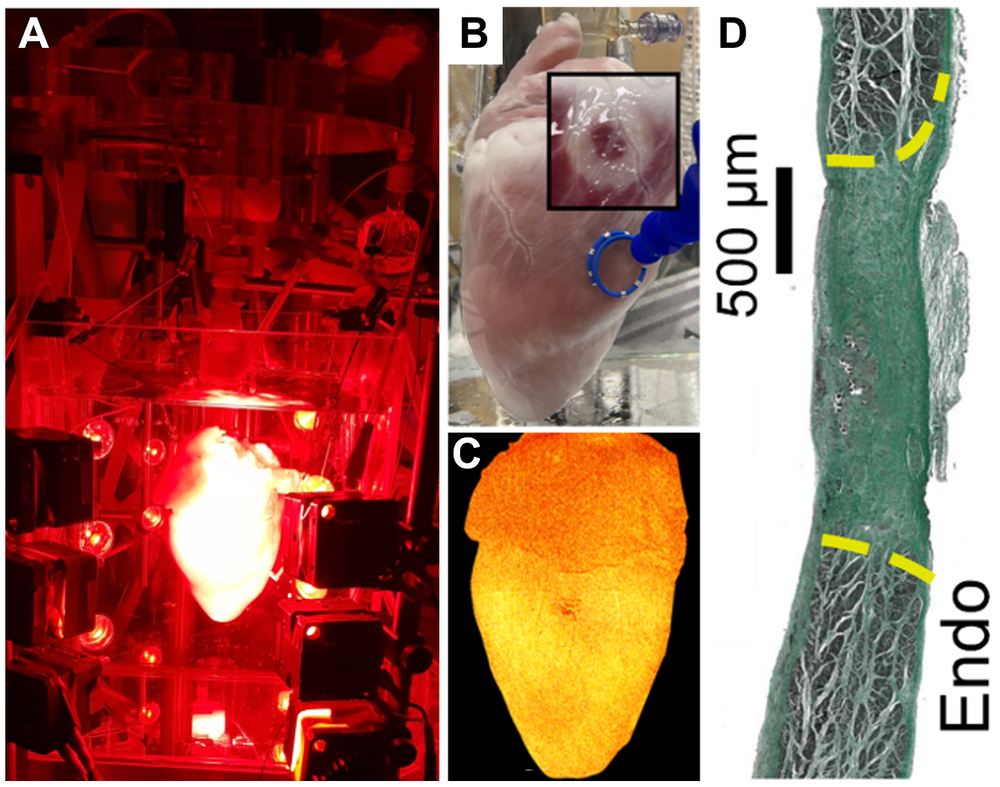
Fig. 2: Ablations performed ex vivo and in vivo.
A) Ablations are performed on a Langendorff-perfused heart while their effects on electrical conduction are monitored using optical mapping.
B) Example of lesions visualised with a live-cell marker (red tissue in the insets indicates metabolically active cells, while white areas indicate the lesion [dead cells]).
C) Image of a heart loaded with a voltage-sensitive dye reporting its electrical activity.
D) Example of a scar obtained after in vivo ablation and recovery, characterised using high-throughput imaging and histological techniques.
Team

Prof. Peter Kohl
Director of IEKM
E-Mail: peter.kohl@uniklinik-freiburg.de

Dr. Rémi Peyronnet
Head of Mechanobiology