4D Imaging / Cardiac NanoDynamics
We use experimental approaches to investigate dynamic cardiac ultrastructure on a nano-to-micro scale. Through our research we attempt to understand how the various cells in the heart respond to their environment, for example to mechanical cues (such as contraction), or the signals generated by their neighbours.
Main research areas are:
Ultrastructure and function of sub-cellular domains in the beating heart
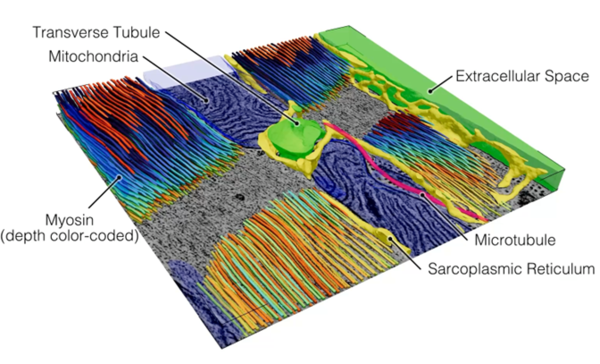
The external appearance of uniformity of cardiomyocyte contractions emerges from coordination of numerous, spatio-temporally heterogeneous processes, involving specialised sub-cellular nano-domains. We investigate the beat-by-beat auto-regulation of heart function at the nano-scale, and propose that the heartbeat is not merely the consequence of cardiomyocyte activity, but an intrinsic regulator that continually modifies nano-domain structure and function in preparation for the next beat. We use time-resolved 3D electron microscopy, sub-cellular functional imaging, ultrastructural proteomics, and artificial intelligence-assisted data analysis, to map out the ultrastructural dynamics of the heart in at the scale of nanometres and milliseconds.
Cardiomyocyte-fibroblast cross talk in the heart
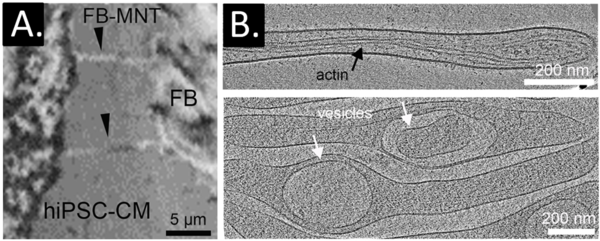
We study the nano-scale cross-talk between cardiomyocytes and fibroblasts in the context of fibrotic remodelling. Specifically, we investigate the role of dynamic fibroblast membrane nanotubes in the process of extracellular matrix deposition at the interface with cardiomyocytes and within cardiomyocyte nano-domains. We use time-resolved light and electron microscopy, human induced pluripotent stem cell (hiPSC) models, and proteomics, to gain insights into nano-scale mechanisms of fibrotic remodelling, and develop strategies to steer fibroblast-cardiomyocyte interactions.
A: Co-culture of primary human cardiac fibroblast (FB, right) and hiPSC-cardiomyocyte (left); fibroblast membrane nanotubes (FB-MNT) directly interact with hiPSC-cardiomyocyte membranes. B: Fibroblast membrane nanotubes can be visualised using 3D cryo-electron tomography. Numerous structures are detected within fibroblast membrane nanotubes, including actin, vesicles, and organelles such as mitochondria.
Cardiomyocyte-neuron cross talk in the heart
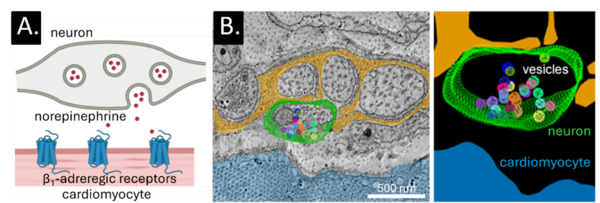
The intracardiac nervous system plays a fundamental role in the fine-tuned regulation of cardiac function. The efficiency and precision of neuro–cardiac communication are thought to be critically determined by the ultrastructural geometry of the interface between neurons and cardiomyocytes. Despite its functional importance, the nano-scale architecture and dynamic organisation of the neuro–cardiac junction remain poorly understood. We investigate the structural organisation and functional properties of the neuro–cardiac junction at the nano-scale, using hiPSC models, and advanced high-resolution imaging approaches.
A: Schematic of a proposed structure of the neuro–cardiac junction, showing a sympathetic neuron bouton forming a close synapse-like approximation with a cardio-myocyte, positioning vesicle release sites directly opposite post-synaptic receptors. B: 3D electron microscopy imaging and reconstruction of unmyelinated nerve fibers in murine sino-atrial node.
- Mechanical deformation of myocyte ultrastructure – structure and function
- Development of novel live fluorescent ion indicators
- Correlative light and electron imaging
- Nano-scale mechanisms of fibrotic remodelling
- Organotypic slice models
- Neuron-cardiomyocyte cross-talk
- Rog-Zielinska EA, Grune J, Kessler T, Lother A, Kohl P. Interstitial cells and arrhythmia. Am J Physiol Cell Physiol 2026/330:C943-C953
- Orós-Rodrigo S, Fu J, Greiner J, Madl J […] Zgierski-Johnston CM [...] Kohl P, Rog-Zielinska EA. Cardiomyocyte mechanical activity counteracts intraluminal calcium depletion in the transverse-axial tubular system during fast electrical stimulation. bioRxiv 2026
- Greiner J, Sonak F, Jones WD, Madl J […] Kohl P, Rog-Zielinska EA. Architecture of the cardiac transverse-axial tubular system across different mammalian species. bioRxiv 2026
- Greiner J, Frangiamore F, Sonak F, Madl J, Seidel T, Kohl P, Rog-Zielinska EA. A deep learning-enabled toolkit for the 3D segmentation of ventricular cardiomyocytes. J Physiol 2025/in press
- Pronto JRD, Mason FE, Rog-Zielinska EA […] Voigt N. Impaired atrial mitochondrial calcium handling in patients with atrial fibrillation. Circ Res 2025/137:1333-1352
- Fu J, Cameron BA […] Schiatti T […] Madl J […] Ravens U, Kohl P, Rog-Zielinska EA, Antoons G, Peyronnet R. Stretch-induced increase in Ca2+-spark rate in rabbit atrial cardiomyocytes requires transient receptor potential ankyrin 1 and intact microtubule network. J Am Heart Assoc 2025/4:e040495
- Greiner J […] Oros-Rodrigo S, Cameron BA, Madl J, Kaltenbacher W, Kok T, Zgierski-Johnston CM, Peyronnet R, Kohl P, Sacconi L, Rog-Zielinska EA. Different effects of cardiomyocyte contractile activity on transverse and axial tubular system luminal content dynamics. J Mol Cell Cardiol 2024/197:125-135
- Paulke NJ […] Lenz C, Rog-Zielinska EA, Lehnart SE, Brandenburg S. Dysferlin enables tubular membrane proliferation in cardiac hypertrophy. Circ Res 2024/135:554-574
- Kohl P, Greiner, J, Rog-Zielinska EA. Electron microscopy of cardiac 3D nanodynamics: form, function, future. Nat Rev Cardiol 2022/19:607-619
- Jünger F [...] Daller CC, Rog-Zielinska EA [...]. 100 Hz ROCS microscopy correlated with fluorescence reveals cellular dynamics on different spatiotemporal scales. Nat Commun 2022/13:1758
- Rog-Zielinska EA, Moss R, Kaltenbacher W, Greiner J, Verkade P, Seemann G, Kohl P, Cannell MB. Nano-scale morphology of cardiomyocyte t-tubule/sarcoplasmic reticulum junctions revealed by ultra-rapid high-pressure freezing and electron tomography. J Mol Cell Cardiol 2021/153:86-92
- Rog-Zielinska EA [...] Peyronnet R, Zgierski-Johnston, Greiner J, Madl J [...] Kohl P. Beat-by-beat cardiomyocyte T-tubule deformation drives tubular content exchange. Circ Res 2021/128:203-215
- MacDonald EA, Madl J, Greiner J […] Rog-Zielinska EA*, Quinn TA*. Sinoatrial node structure, mechanics, electrophysiology and the chronotropic response to stretch in rabbit and mouse. Front Physiol 2020/11:908 (*joint authorship)
- Kong CHT*, Rog-Zielinska EA*, Kohl P, Orchard CH, Cannell MB. Solute movement in the t-tubule system of rabbit and mouse cardiomyocytes. PNAS USA 2018/115:E7073-E7080 (*joint authorship)
- Rog-Zielinska EA, O'Toole ET, Hoenger A, Kohl P. Mitochondrial Deformation During the Cardiac Mechanical Cycle. Anat Rec 2018/302:146-152
- Scardigli M [...] Rog-Zielinska EA, Kohl P [...] Sacconi L. Quantitative assessment of passive electrical properties of the cardiac T-tubular system by FRAP microscopy. PNAS USA 2017/114:5737-5742
- Brandenburg S [...] Rog-Zielinska EA [...] Kohl P [...] Lehnart SE. Axial tubule junctions control rapid calcium signaling in atria. J Clin Invest 2016/126:3999-4015
- Quinn TA*, Camelliti P*, Rog-Zielinska EA* [....] Kohl P. Electrotonic coupling of excitable and non-excitable cells in the heart revealed by optogenetics. PNAS USA 2016/113:14852-14857 (*joint authorship)
- Rog-Zielinska EA, Johnston CM [...] Kohl P. Electron tomography of rabbit cardiomyocyte three-dimensional ultrastructure. Prog Biophys Mol Biol 2016/121:77-84
- Rog-Zielinska EA, Norris RA, Kohl P, Markwald R. The Living Scar - Cardiac Fibroblasts and the Injured Heart. Trends Mol Med 2016/22:99-114
Team

Eva Rog-Zielinska, PhD
Head of section

Dr. Josef Madl
Senior Scientist
E-Mail: josef.madl@uniklinik-freiburg.de

Hannah Kappler

Sofía Orós Rodrigo

Patricia Aparecida Morais Costa

Wenzel Kaltenbacher

Wesley Dean Jones

Nina Steinhagen

Eric Suaudeau

Jana Meyer
E-Mail: jana.meyer@uniklinik-freiburg.de

Jaide Cashin