Flow Cytometry
Cell SortingBooking of Cell Sorters
- Appointment requests should be sent via email to facslab@uniklinik-freiburg.de.
- Registred and experienced users can use our booking calendar “PPMS” booking system.
- Standard sorting times are Monday to Thursday between 9:00 and 16:30 and on Friday between 9:00 and 14:30.
- Please specify the following information in your request
- Desired date / time
- Duration of sort
- Desired cell-sorter (MoFLo, Aria, Fusion)
- Nozzle Size (70, 85, 100, 130 µm). If not specified, the 70 µm Nozzle is used by default
- Origin of the cells (human, mouse, cell culture etc.)
- Cell number / sample size and number of samples
- Biosafety-Level (S1,2,3**)
Cell Sorting Service Fee
The costs for cell sorting for academic users are 50 €/hour + 25 €/appointment. The billing cycle includes both the actual sort time as well as time required for setting up the experiment. Billing begins with the start of the booked appointment. If appointments are canceled upon short notice and it is not possible to find an alternative appointment to fill the slot, 50% of the originally booked time will be charged.
Sorters
Lighthouse operates three high speed cell sorters: a 16-color FACS Aria Fusion and a 17-color FACS Aria III, both from Becton Dickinson, as well as a 14-color MoFlo Astrios cell sorter from Beckman Coulter as well as a Cytoflex SRT with 15 colors. Due to their complexity these machines must be operated by members of the Core Facility. Note: Because the sorters are heavily booked (wait times are often 3 weeks), it is best to reserve times on the instruments as early as possible. If you need more information as to which combinations of dyes or fluorescent proteins it is possible to use, please contact us. The best time to do this is when first planning a new experiment.
Lasers Available - Cell Sorters
|
FACS Aria Fusion MoFlo Astrios Cytoflex SRT |
FACS AriaIII |
Fluorochrome Examples |
|---|---|---|
|
Near UV: 375 nm* *colinear with 405 nm laser |
DAPI, Hoechst (including detection of SP cells) |
|
|
405 nm |
405 nm |
DAPI, Alexa 405, CFP, Brilliant Violet Dyes, Pacific Blue, Live/Dead Aqua, Zombie Aqua, etc. |
|
488 nm |
488 nm |
FITC, Alexa 488, Brilliant Blue 515, GFP, YFP, CFSE PerCP, PerCP-CY5.5 |
|
561 nm |
561 nm |
PE, PE-Texas Red, PE-CY5, PE-CY5.5, PE-CY7 dsRed, tdTomato, mCherry, etc. |
|
638/640 nm |
633 nm |
APC, Alexa 633/647, TO-PRO3, APC-CY7, APC-H7, Alexa 680/700, etc. |
Other possibilities for the cell sorters are:
- Cell/well cloning directly onto slides or into i.e. 96- or 384-well tissue culture plates (up to 1536 Wells on the MoFlo Astrios)
- 4-way/6-way sorting: Up to 4 populations can be sorted simultaneously on the Arias and up to 6 populations on the MoFlo Astrios
- Index Sorting - the ability to look up the phenotype of a cell after cloning
- Small particle detector - MoFlo Astrios EQ
- Biosafety Hood - FACS Aria Fusion
For more information about setting up your samples for sorting and the best buffers to use, with your samples please also see: Sample Preparation
Sample tubes
Because of their differing electrostatic properties, it is recommended that you use polystyrene tubes (clear) as sample tubes, and polypropylene tubes (opaque) for sample collection, Sample tubes should be 12 x 75 mm in size, while collection tubes can be either 12 x 75 mm in size, 15 ml conical tubes for large samples, or 1.5 ml tubes for small samples.
Cloning into Plates
When cloning into plates, be sure to plate out the wells with a large amount of medium, so that they are ~75% full. This makes it easier to sort into the well, with less chance of the droplet landing on the side of the well. For special plates or formats it is helpful to bring by an empty plate to set up the stage positions ahead of time.
How to Select the Correct Nozzle Size
As a general rule of thumb: The cell size should not exceed one-fifth of the nozzle diameter. Using the wrong nozzle size causes fanning of the side streams, poor sort purity and nozzle clogging.
|
|
Nozzle Size |
70 µm |
85 µm (Arias) |
100 µm |
130 µm (Arias) |
|---|---|---|---|---|
|
Pressure (pounds per sq. in.) |
60-70 psi |
45 psi |
20-25 psi |
10 psi |
|
Max. Cell Diameter |
14 µm |
17 µm |
20 µm |
26 µm |
|
Max. Events/Sec. |
22.000 - 30.000 |
11.000 |
7.000-10.000 |
3.000 |
|
Max. Events/Hour |
7-9x10e7 |
3-4x10e7 |
2x10e7 |
1x10e7 |
|
Starting Cell Conc. of Sample |
3-5x10e7/mL |
1-2x10e7/mL |
5-7x10e6/mL |
2-4x10e6/mL |
|
Approx. Size of Drop |
1 nL |
2 nL |
4 nL |
11 nL |
|
Cell Concentration Post-Sort |
1x10e6/mL |
0,5x10e6/mL |
2,5x1065/mL |
1.1x10e5/mL |
If you are unsure which nozzle size you will require, just ask one of the sort operators and they will be happy to advise you. They can also help you to determine what diameter your cells have while in suspension.
For additional help with panel design, the configurations of all Lighthouse sorters and analysers are now listed on the Fluorofinder website. To access information about the cell sorters please follow the links below.
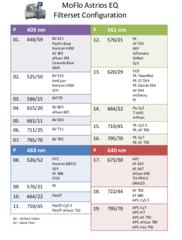
MoFlo Astrios Lasers and Channels
Overview about lasers and suitable fluorochromes of cell sorter ARIA III.
FUSION Lasers & Channels
Overview about lasers and suitable fluorochromes of cell sorter ARIA FUSION.
ARIA III Lasers and Channels
Overview about lasers and suitable fluorochromes of cell sorter ARIA III.
Beckman Coulter Cytoflex SRT Lasers and Channels
Overview about lasers and suitable fluorochromes of cell sorter Cytoflex SRT
Biosafety
The cell sorters normally operate at pressures of 60 - 70 psi, and can aerosolize a portion of the sample. The Lighthouse cell sorting service is classified to a Biosafety Level of 2. Therefore only cells of risk groups 1 and 2 can be sorted. .
It is necessary to determine the risk potential of the cells prior to sorting to take appropriate precautions. For this reason, please fill out the following form.
Note:
It is not necessary to fill out the checklist for each individual Sort. However, it must be filled out for each different cell type, treatment and origin of the cells.
Sort experiments of any biosafety level can only be carried out after filling out the proper form. All new projects must be registered with this form. It can be found in our "Download area".
FACS Analysis
For information about our FACS Analysers please see the FACS Analysis page.
Offline Analysis
Cloud Licenses:
Lighthouse now has a FluoroFinder Academic Plus Sitewide License. Fluorofinder has many useful tools to help you get started with panel design, in addition to a spectra viewer, validated panels, and directories of antibodies and fluorophores. All Lighthouse cytometers (analyzers and sorters) are listed on Fluorofinder, to make it easier for our users to develop panels for their experiments. Access for all is free, with more advanced tools available under the Sitewide license (for example AI Intellipanel tool). If you would like access to the Lighthouse Academic Plus Sitewide license, please contact us at FACSlab@uniklinik-freiburg.de and we will have your account activated.
Lighthouse also has a Cytobank Premium Cloud License, with many tools for dimensionality reduction, correction of batch effects and other useful tools for high-end cytometry analysis. All Lighthouse users can use the license. To get access to Cytobank, please contact us at FACSlab@uniklinik-freiburg.de and we will have your account activate.
The Core Facility also has offline analysis workstations with the flow cytometry software programs FlowJo and Kaluza available for general use.