Low-Field MR Technology
Low-field NMR has received increasing attention in recent years, especially with respect to nuclear hyperpolarization [1–8]. The combination of both methods seems appealing because: (i) hyperpolarization (over) compensates lacking signal at low field [9–16], (ii) some hyperpolarization techniques necessitate low-field NMR for spin-order transfer (SOT) [17–19], and (iii) low-field MR systems are much less hardware-demanding and cost-intensive than high-field systems. The combination of these technologies holds great potential for biomedical research, including in vivo imaging and detection of targeted markers, but without the need of superconducting magnets and extensive infrastructure. Entirely new applications like portable diagnostic devices may emerge. However, only a few low-field NMR systems have been described, each tailored to a very specific application, e.g. hyperpolarized gases [4, 5], prepolarized solutions [2, 3] or a relatively high field [20].
Most reports describe signal detected from samples with large, non-equilibrium polarization. No detection of thermal 1H signal was reported below 4.5 mT [7]; 1 6 mT, though, is exactly the range where most pH2-based 13C - hyperpolarization experiments were carried out [17–19, 21–23], and the highest signal amplification by reversible exchange (SABRE) was predicted and demonstrated [24–26]. A dedicated NMR detection system to aid pH2-based hyperpolarization experiments at 1 - 6 mT was not yet described. The requirements for such a device to perform PASADENA (Parahydrogen and synthesis allows dramatically enhanced nuclear alignment) / PHIP (parahydrogen induced hyperpolarization) [17, 27–29] are the following (a-c):
(a) Both methods require a homogeneous, low magnetic field (B0) e.g. 1.8 mT or 6 mT for effective hyperpolarization. It was shown that low-field spin order transfer 13C hyperpolarization suffers from B0 instability [22], and SABRE is strongly dependent on the field strength [24, 25]. (b) To transfer the spin order from pH2 to another nuclei at low field [17, 18, 30], exact manipulations of 1H and X nuclei is needed. Inaccurate B1 decreases the polarization yield and may cause complete loss, as was shown in theory [31] and experimentally [22, 23]. (c) Previously, a sample was hyperpolarized at low field and transported to a high field magnet for quantification in tens of seconds [19, 21, 22, 24]. The great range of magnetic fields encountered during transport, however, disturbs the quantification and may annihilate the polarization completely [32]. These issues are avoided by direct detection and quantification of a hyperpolarized sample at low field. For absolute quantification, detection of hyperpolarized samples as well as sufficient sensitivity for samples in thermal equilibrium at low field is required.
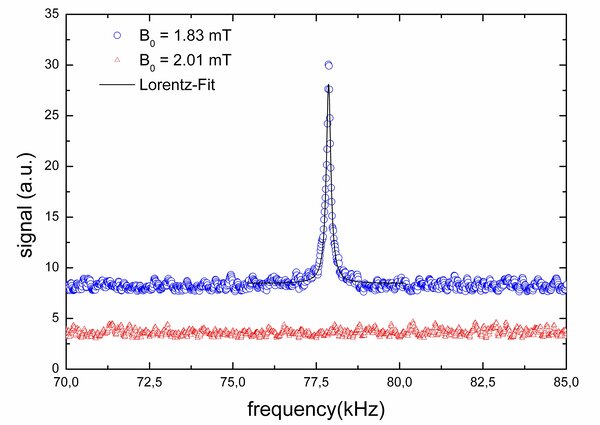
Figure 1: 1H-NMR magnitude spectrum after 100 pulse - acquisition experiments of water (blue circles, top). To verify the origin of the resonance, the experiment was repeated at B0 = 2 mT (f(1H) = 100 kHz), keeping all other parameters constant, including the excitation pulse at 79 kHz. Consequently, no resonance was observed (red triangles, bottom), thus confirming the NMR origin of the signal (90° pulse at 6.8 V and 397 us, TR = 5 s, acquisition time 25 ms, acquisition delay 1.5 ms).
Apart from hyperpolarization, low-field NMR offers insight into low-frequency MR physics and may be used for educational purposes, as such devices provide a much closer access to the underlying physics and are much more cost effective when it comes to hands-on training.
The aim of this project is three-fold: to advance low-field MR technology, to enable thermal mT NMR and to aid pH2-based hyperpolarization. Specifically, we designed a low-field NMR apparatus based on a transmit-only pH2-polarizer [18, 23] which
1. is suitable for NMR of ul to a few ml solution volume at low mT fields, 2. allows precise manipulations of the spins i.e. flip angle, 3. is sufficiently sensitive and flexible for moderately complex low-field NMR experiments (pulses of arbitrary frequency and shape followed by a detection interval), 4. is easy to construct, to reproduce and cost-effective, 5. is suited for educational purposes and 6. addresses the needs of parahydrogen based hyperpolarization methods as laid out above.
Dr. Andreas B. Schmidt
Head of Hyperpolarization
Tel.: +49 761 270-93880
E-Mail: andreas.schmidt@uniklinik-freiburg.de
University Medical Center Freiburg
Dept. of Radiology · Medical Physics
Killianstr. 5a
79106 Freiburg

